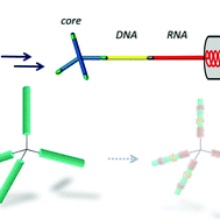
By combining both chemical and enzymatic ligation with procedures guiding the self-assembly of nanotubular tobacco mosaic virus (TMV)-like particles (TLPs), novel nucleoprotein structures based on DNA-terminated branching elements, RNA scaffolds and TMV coat protein (CP) are made accessible. Tetrahedral tetrakis(hydroxybiphenyl)adamantane cores with four 5′-phosphorylated dinucleotide arms were coupled to DNA linkers by chemical ligation. The resulting three-dimensional (3D) branching elements were enzymatically ligated to the 3′ termini of RNA scaffolds either prior to or after the RNAs’ incorporation into TLPs. Thus, architectures with interconnected nanotube domains in two different length classes were generated, each containing 70 CP subunits per 10 nm length. Short TMV origin-of-assembly-containing RNA scaffolds ligated to the DNA allowed the growth of protein-coated 34 nm tubes on the terminal RNA strands in situ. Alternatively, 290 nm pre-fabricated tubes with accessible RNA 3′ termini, attained by DNA blocking elements hybridized to the RNAs, were ligated with the branched cores. Both approaches resulted in four-armed nanoobjects, demonstrating a so far unique combination of organic synthesis of branching elements, enzymatic modifications, nucleic acid-based scaffolding and RNA-guided and DNA-controlled assembly of tubular RNA-encapsidating protein domains, yielding a novel class of 3D nucleoprotein architectures with polyvalent protein elements. In the long term, the production route might give rise to supramolecular systems with complex functionalities, installed via the orthogonal coupling of effector molecules to TLP domains.