The amygdala, a brain structure in the temporal lobe, is a key structure for storing fear memories. Fear memories can be modified by a second learning process called extinction, which is the basis for behavioral therapies in the treatment of anxiety disorders. Here, the individual learns that certain stimuli are not fearful anymore in a specific setting. Extinction depends on a larger brain network comprising the amygdala, the hippocampus and the medial prefrontal cortex, as well as interactions between them. To date, some of the strongest links between neural plasticity and behavioral learning come from studies of fear memory in the amygdala. Changes can be detected ex vivo in recordings from brain slices obtained from trained animals. Our research hinges on using this approach. While plastic changes of sensory inputs to excitatory cells in the amygdala are well understood, inputs from other structures and intra-amygdala inhibitory elements may also undergo plasticity. Our goal is to identify and investigate novel changes in these networks.
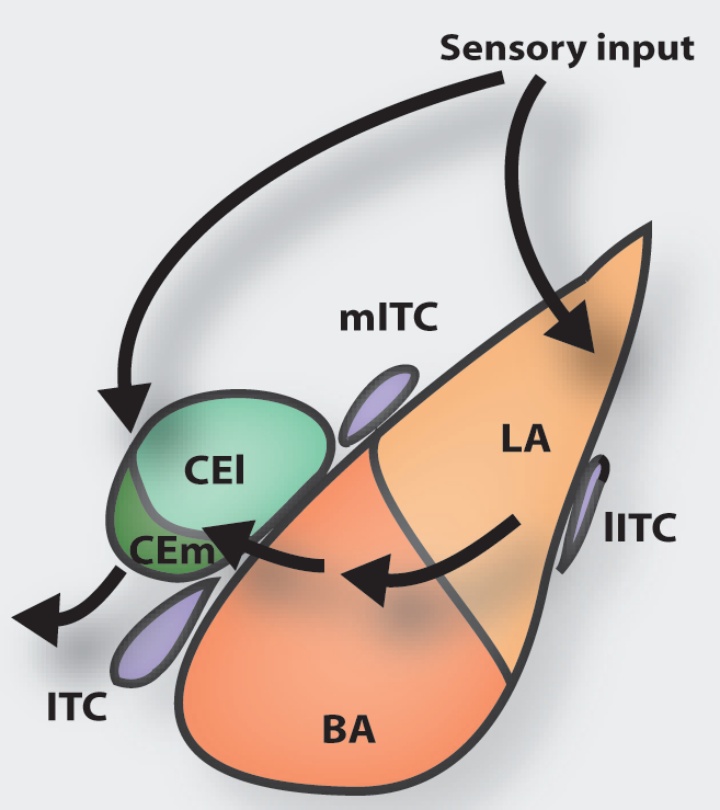
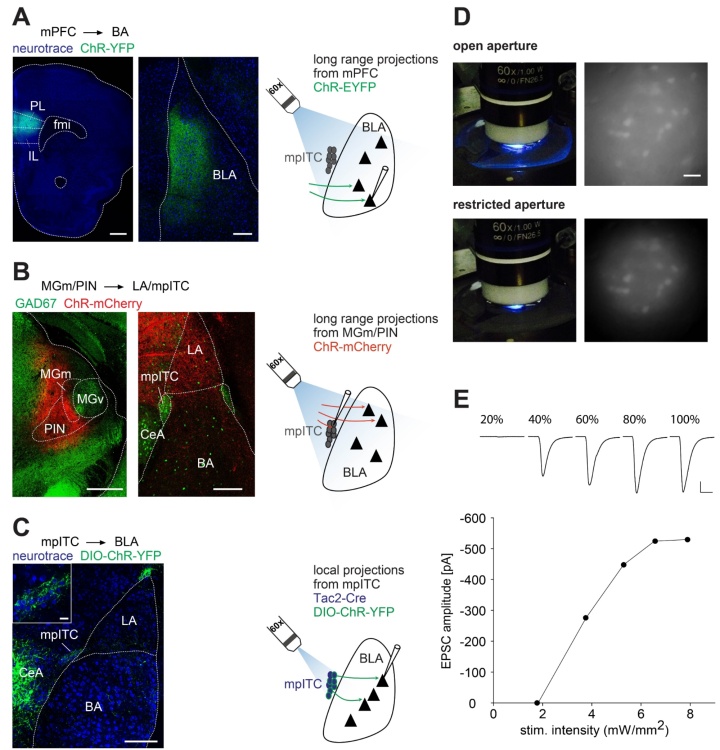
One line of research aims to understand properties and function of a specialized group of inhibitory neurons in the amygdala, the intercalated cells. These cells have recently received much attention, as they become activated during fear behaviors and are critical for extinction. However, little is known about their properties and connectivity. We combine electrophysiological, optogenetic and anatomical techniques with behavioral manipulations to understand how they are integrated and process information within amygdala networks and may participate in shaping acquired fear behavior and beyond. Moreover, we are interested elucidating the role of neuromodulatory inputs and processes to this distinct network of neurons.
A second line of research investigates extinction mechanisms. On the one hand, we are interested in understanding the cellular interactions within extinction networks, which are critical for understanding extinction mechanisms. Towards this end, we use tracing, reporter mice and ex-vivo optogenetic approaches that allow for targeted stimulation of long-range inputs to specific cells in the amygdala. On the other hand, we investigate systemic processes that modulate consolidation of extinction memory and influence these circuits. In that respect, we explore the idea that sleep, which has been show to be beneficial for many forms of memory, also supports fear extinction.
We also collaborate with other researchers on sleep-related cellular and plasticity mechanisms in the brain.
A third line of research addresses developmental processes in amygdala circuits that could underlie developmental differences in learning behavior. The ability to learn fear first emerges in infancy and changes into adulthood. Extinction learning emerges in juveniles, but is different from adults. We used electrophysiological approaches investigating developmental changes particularly in amygdala circuits between infancy and adulthood. Furthermore, we are interested in applying manipulations to understand the function of specific developmental processes in amygdala plasticity and learning.

Ingrid Ehrlich
Prof. Dr.Head of Department
[Photo: Patrick Münster]