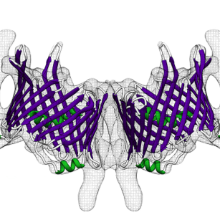
Summary:
The TOM complex is the main entry gate for protein precursors from the cytosol into mitochondria. We have determined the structure of the TOM core complex by cryoelectron microscopy (cryo-EM). The complex is a 148 kDa symmetrical dimer of ten membrane protein subunits that create a shallow funnel on the cytoplasmic membrane surface. In the core of the dimer, the β-barrels of the Tom40 pore form two identical preprotein conduits. Each Tom40 pore is surrounded by the transmembrane segments of the α-helical subunits Tom5, Tom6, and Tom7. Tom22, the central preprotein receptor, connects the two Tom40 pores at the dimer interface. Our structure offers detailed insights into the molecular architecture of the mitochondrial preprotein import machinery.
Highlights:
- Cryo-EM reveals structure of the mitochondrial preprotein translocase TOM
- Two identical β-barrels form a twin pore across the outer membrane
- Two long α helices of the preprotein receptor Tom22 hold the dimer together
- The small α-helical subunits Tom5, Tom6, and Tom7 surround the twin pore
For reference, see Bausewein M., Mills D., Langer J., Nitschke B., Nussberger S. & Kühlbrandt, W. Cell 170: 693–700 (2017)